Factor XIa inhibition with asundexian after acute non-cardioembolic ischaemic stroke (PACIFIC-Stroke): an international, randomised, double-blind, placebo-controlled, phase 2b trial - The Lancet
Por um escritor misterioso
Last updated 03 março 2025


Baseline Characteristics According to Treatment Group in

Design and Preclinical Characterization Program toward Asundexian
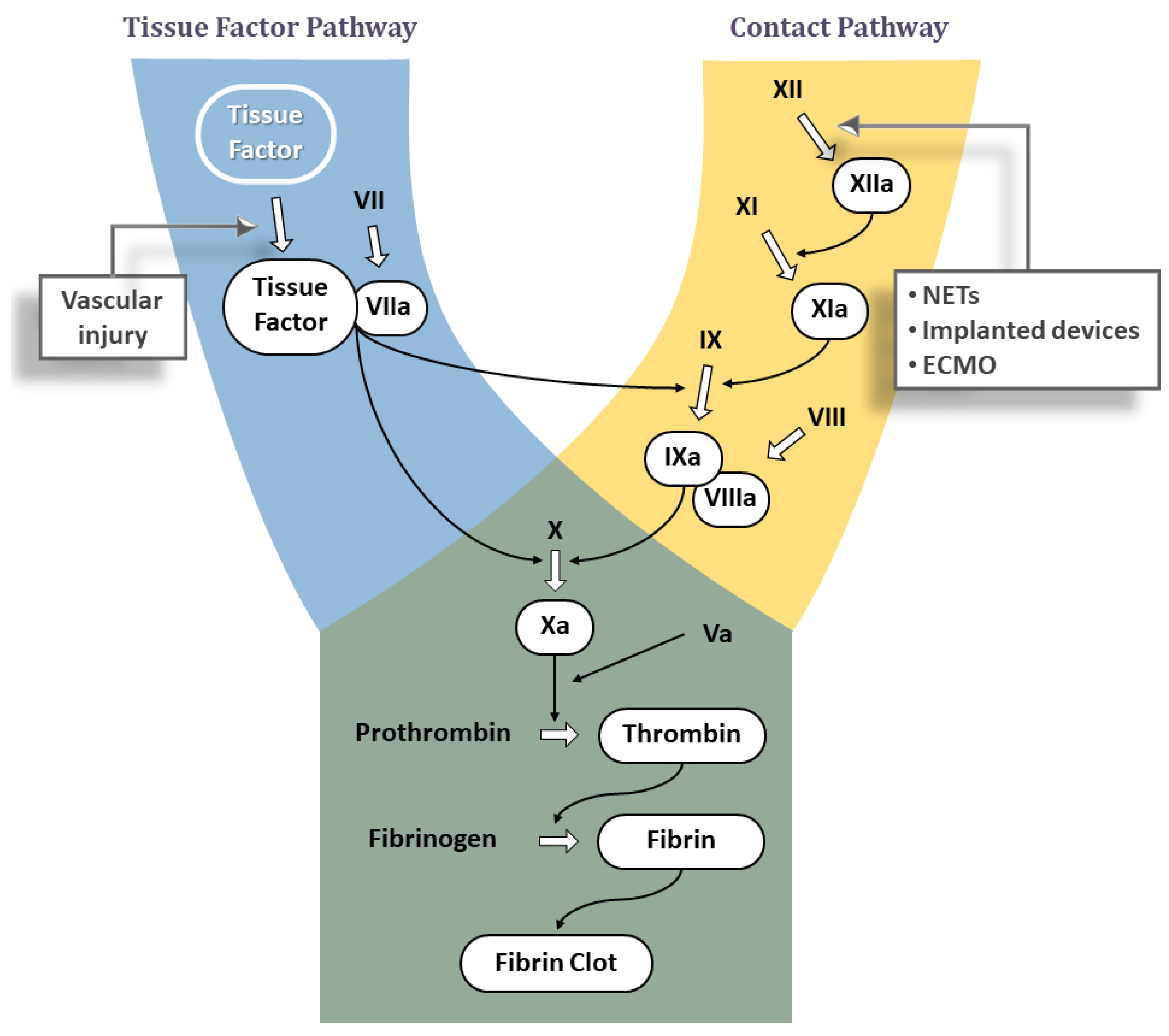
JCDD, Free Full-Text

Antithrombotic Therapy for Primary and Secondary Prevention of

Factor XIa inhibition with asundexian after acute non
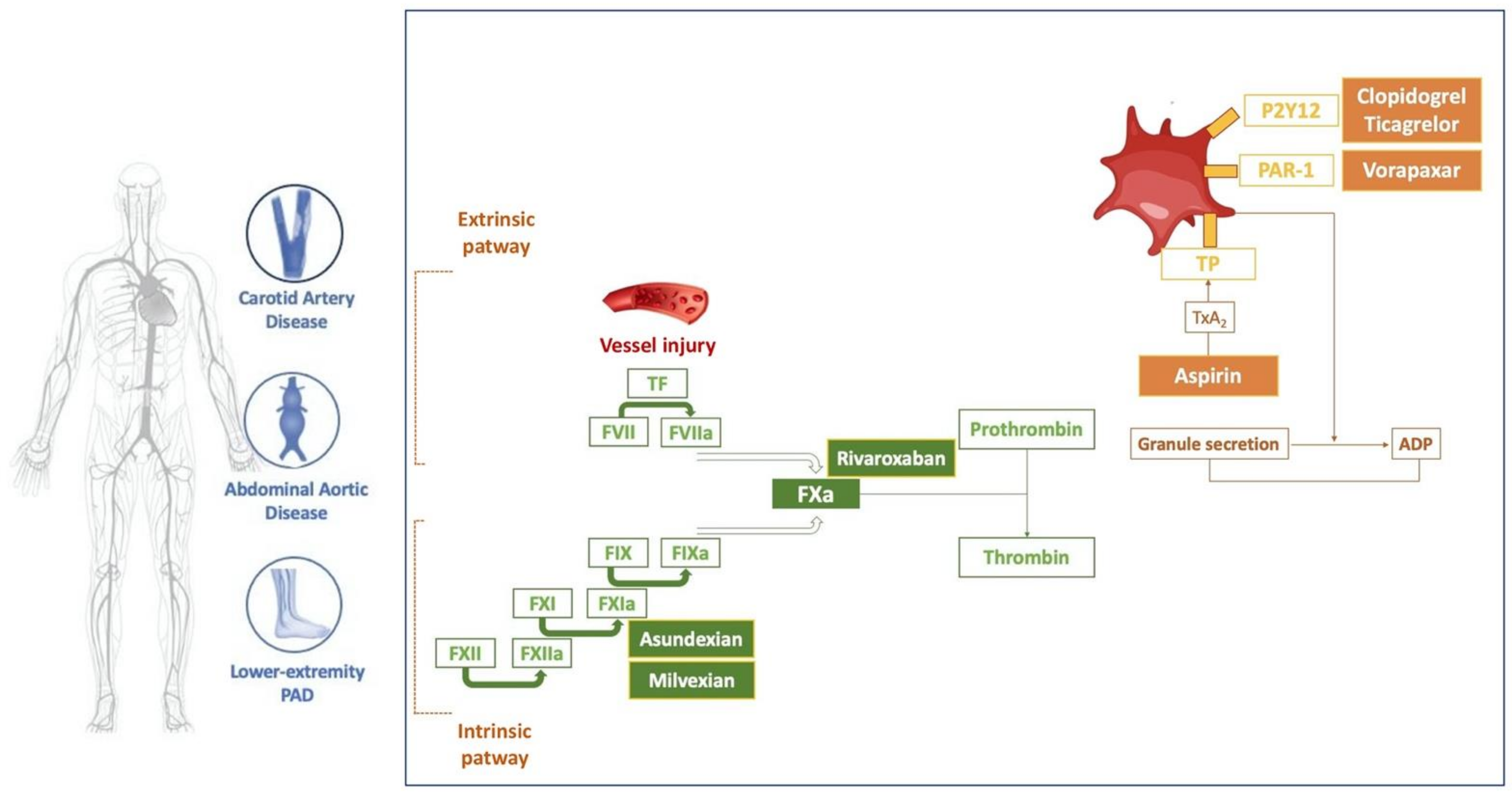
JCDD, Free Full-Text

PDF) Pharmacological targets of Asundexian relevant to its

Clinical Evaluation of Factor XIa Inhibitor Drugs: JACC Review

Ischaemic stroke despite antiplatelet therapy: Causes and outcomes
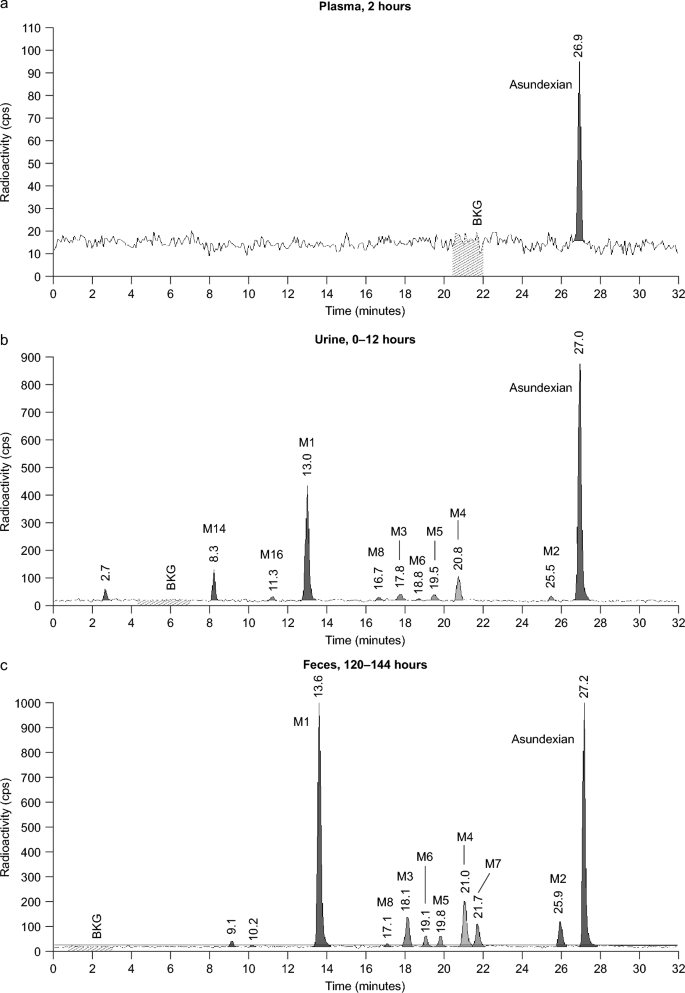
Metabolism and Disposition of the Novel Oral Factor XIa Inhibitor

Frequency and Patterns of Brain Infarction in Patients With

ESC 22: Dosing of Asundexian in Pts With Non-Cardioembolic

Asundexian (BAY-2433334), CAS 2064121-65-7
Recomendado para você
-
 Brain Test Level 372 He wants big muscles in 202303 março 2025
Brain Test Level 372 He wants big muscles in 202303 março 2025 -
brain test level 372|TikTok Search03 março 2025
-
 Brain Test Level 202 Solve the puzzle in 202303 março 2025
Brain Test Level 202 Solve the puzzle in 202303 março 2025 -
 Close this dialog03 março 2025
Close this dialog03 março 2025 -
 Easy Game Brain Test Level 372 Finish shopping.03 março 2025
Easy Game Brain Test Level 372 Finish shopping.03 março 2025 -
The Hop Shoppe03 março 2025
-
 Microbiota‐gut‐brain axis as a regulator of reward processes - García‐Cabrerizo - 2021 - Journal of Neurochemistry - Wiley Online Library03 março 2025
Microbiota‐gut‐brain axis as a regulator of reward processes - García‐Cabrerizo - 2021 - Journal of Neurochemistry - Wiley Online Library03 março 2025 -
Introduction to Cortical Neurons03 março 2025
-
 Atlanta Pediatric Research, Emory + Children's + GT03 março 2025
Atlanta Pediatric Research, Emory + Children's + GT03 março 2025 -
 A sense of self03 março 2025
A sense of self03 março 2025
você pode gostar
-
 Camiseta Luffy - One Piece03 março 2025
Camiseta Luffy - One Piece03 março 2025 -
 Leadale no Daichi nite Anime characters, Anime, Character03 março 2025
Leadale no Daichi nite Anime characters, Anime, Character03 março 2025 -
 Téléchargement de l'application Gênio Quiz HP 2023 - Gratuit - 9Apps03 março 2025
Téléchargement de l'application Gênio Quiz HP 2023 - Gratuit - 9Apps03 março 2025 -
 Xbox Game Pass Ultimate chega ao Android com mais de 150 jogos disponíveis; confira lista03 março 2025
Xbox Game Pass Ultimate chega ao Android com mais de 150 jogos disponíveis; confira lista03 março 2025 -
Club Atlético Independiente de Burzaco - Fronte Ader Valentino, Cremona Tobias, Lobato Martinez Micaela Oriana, Entrenadora Gisela Dadona, representaran al C.A.I. Burzaco en los Juegos Bonaerenses en la etapa Municipal a desarrollarse03 março 2025
-
 BONA-Sapatos esportivos masculinos duros, tênis de corrida ao ar livre, calçados pretos, todos os jogos03 março 2025
BONA-Sapatos esportivos masculinos duros, tênis de corrida ao ar livre, calçados pretos, todos os jogos03 março 2025 -
 Barakamon Barakamon, Anime love, Anime03 março 2025
Barakamon Barakamon, Anime love, Anime03 março 2025 -
 Amos Bocelli on Instagram: “An instant of a unique night. Thanks03 março 2025
Amos Bocelli on Instagram: “An instant of a unique night. Thanks03 março 2025 -
 Pochita: Tudo sobre o cachorrinho motosserra de Chainsaw Man03 março 2025
Pochita: Tudo sobre o cachorrinho motosserra de Chainsaw Man03 março 2025 -
 Pólipo endometrial: Dr. Rubens do Val responde! - Clínica Rubens do Val CRM 5876403 março 2025
Pólipo endometrial: Dr. Rubens do Val responde! - Clínica Rubens do Val CRM 5876403 março 2025


